For decades, Alzheimer’s disease has been called one of medicine’s greatest unsolved puzzles. Scientists have studied genetics, lifestyle, and environmental triggers in search of answers.
But a growing body of research now points to a suspect hiding in plain sight — fungi/molds.
Fungi/Molds are everywhere.
They live in our homes, our gut, and on our skin. Most of the time, they cause no harm.
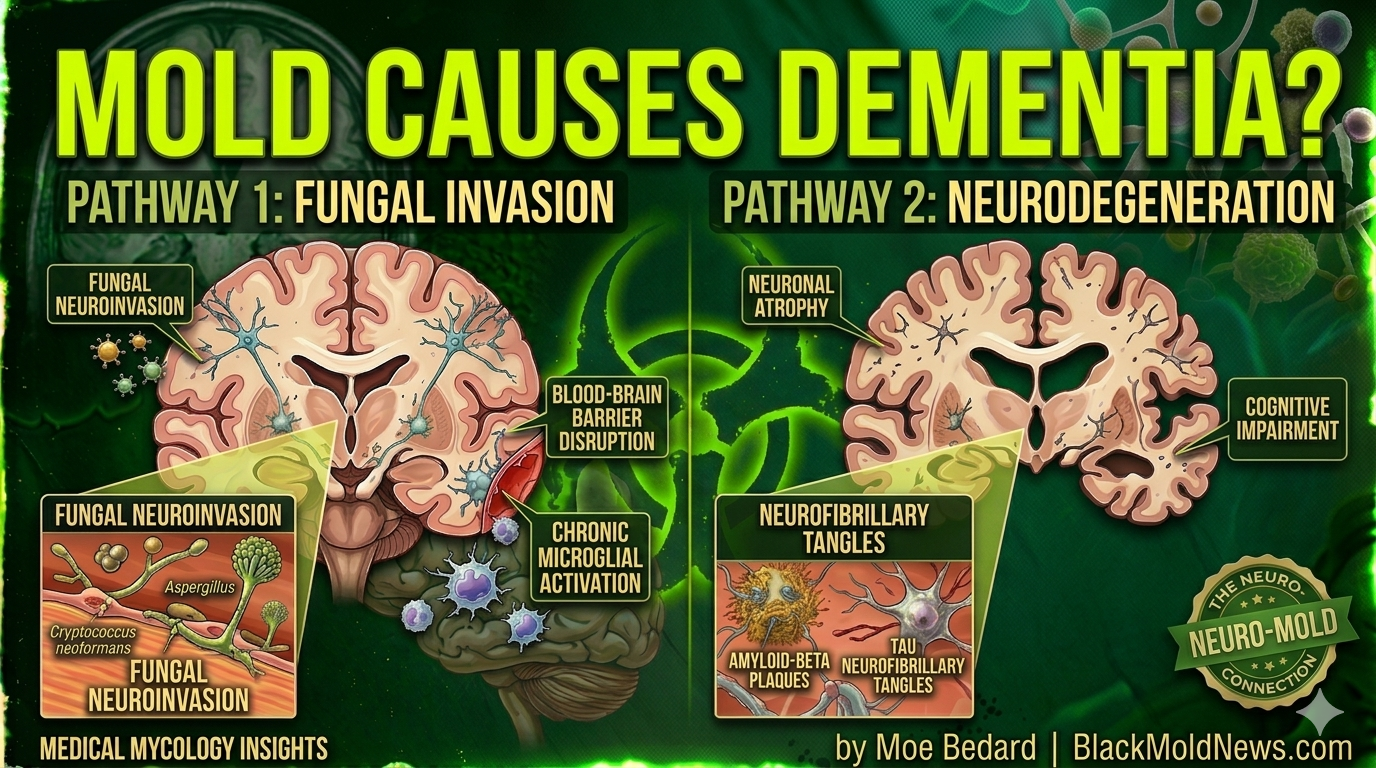
But when fungal organisms breach the blood-brain barrier and take hold in brain tissue, the consequences may be devastating.
Emerging evidence suggests that fungal infections — and the toxic compounds they produce called mycotoxins — can trigger the very chain of events that define Alzheimer’s disease: inflammation, amyloid plaque buildup, neuron death, and cognitive decline.
This isn’t fringe science.
It’s being published in peer-reviewed journals, studied at major university labs, and funded by organizations like the Infectious Diseases Society of America.
For homeowners living with mold and patients struggling with cognitive problems, this research may represent a major turning point in how we understand brain disease.
The most striking evidence came in 2015, when a Spanish research team published a landmark study in the journal Scientific Reports. They examined brain tissue samples from 11 Alzheimer’s patients and a control group of healthy individuals.
The findings were dramatic.
Every single Alzheimer’s patient tested showed fungal material in multiple brain regions — including the frontal cortex, cerebellum, entorhinal cortex, hippocampus, and even blood vessels supplying the brain.
The control subjects showed no such fungal/mold contamination.
The fungal species identified included Alternaria, Botrytis, Candida, Malassezia, Cladosporium, and Cryptococcus.
These are not exotic tropical organisms.
Several of them are common household molds. Cladosporium and Alternaria are routinely found in water-damaged homes. Candida is a yeast that most people carry in their gut at low levels.
What made the Alzheimer’s brains different was the burden of infection.
The fungal load in AD patients was significantly higher than in elderly control subjects, and the species composition was clearly distinct.
This was not background contamination.
These were active f infecungaltions embedded in living neural/brain tissue.
The authors of the study stated plainly:
“Our findings provide compelling evidence for the existence of fungal infection in the CNS from AD patients, but not in control individuals.”
In 2018, a follow-up study published in Frontiers in Aging Neuroscience confirmed these findings and further stated:
“We have provided extensive evidence that disseminated mycoses are implicated as causative agents or as risk factors for AD”.
Fungal components — proteins, DNA, and cellular structures — were consistently identified in post-mortem AD brain tissue using immunohistochemistry, PCR analysis, and proteomic profiling.
A landmark 2023 study from Baylor College of Medicine discovered exactly how a common fungus can trigger Alzheimer’s-like damage in the brain.
Researchers found that Candida albicans produces enzymes called secreted aspartic proteinases (Saps) that actively break down the blood-brain barrier, allowing the fungus to invade brain tissue.
Once inside, the fungus triggers production of amyloid-beta–like peptides — the same toxic fragments associated with Alzheimer’s disease plaques. As the lead researcher Dr. Yifan Wu explained:
“We found that Candida albicans produces enzymes that break down the blood-brain barrier, giving the fungus access to the brain where it causes damage.”
The authors concluded that C. albicans is “linked to chronic brain diseases such as Alzheimer’s disease” and that the brain’s immune response to the fungus mirrors the same inflammatory mechanisms seen in AD.
A 2023 review in IMR Press’s Journal of Integrative Neuroscience documented how specific mold toxins cause structural brain damage that overlaps directly with Alzheimer’s pathology. The review found that:
“Inflammation stimulated by mycotoxins causes inflammatory markers such as NF-kappa B and TNF-alpha to access the OB and frontal cortex, leading to the deposition of amyloid-beta plaques — an Alzheimer’s disease pathological hallmark.”
The same study found that Satratoxin (produced by Stachybotrys chartarum, i.e., black mold) causes olfactory nerve apoptosis and bilateral atrophy of the olfactory bulb in the brain.
A 2025 paper in ScienceDirect specifically examined mycotoxin exposure as “a potential etiological factor for neurodegenerative diseases” and confirmed that compounds like T-2 toxin and deoxynivalenol (DON) can breach the blood-brain barrier and infiltrate brain tissue.
The study noted that fungal presence has been documented in brain tissues of both Alzheimer’s and Parkinson’s patients.
An earlier A 2003 study titled, “How a Slime Mold Came to the Aid of Alzheimer’s Research,” details how a structure called a “Hirano body” found in a slime mold is present in increased amounts in Alzheimer’s patients.
It is mostly present in the brain’s major site of learning and memory, the hippocampus.
The study reports, “What causes neurodegenerative diseases like the most common early symptom of Alzheimer’s which is difficulty remembering newly learned information because Alzheimer’s changes typically begin in the part of the brain that affects learning, is still largely unknown, but something destroys nerve cells in the brain over a period of time as victims gradually lose their minds.”
According to the study, “The researchers stated that since then Hirano bodies have been reported in the brains of people with neurodegenerative diseases, as well as diabetes, stroke and alcoholism.
They said:
“Making a Hirano body may be a cellular mechanism for dealing with run-amuck proteins,” said Furukawa, who manages the day-to-day operation of the lab. “That’s just a hypothesis.”
Run-amuck proteins certainly seem to play some role in all the diseases where Hirano bodies are found.
“Perhaps Hirano bodies do nothing; perhaps they’re part of cell death; or perhaps they are adaptations to stress that are good for cells,” Fechheimer said.
“Because they’re seen in so many diseases, it’s worth finding out.”
The researchers concluded;
“Our findings grew out of research funded by the National Science Foundation,” said Fechheimer, who also received support from the Alzheimer’s Association once the link to neurodegenerative disease was established. “That’s why NSF is very excited by this connection to something that sounded like brain disease. To them it was a wonderful example why you never want to forget about investing in basic research.”
How Fungi Get Into the Brain
The brain is protected by the blood-brain barrier (BBB) — a tightly sealed wall of specialized cells that controls what enters the brain from the bloodstream. It is one of the most selective barriers in the human body.
So how do fungi get through?
Researchers at Baylor College of Medicine identified a key mechanism in a 2023 study published in the journal Cell Reports. They found that Candida albicans produces enzymes called secreted aspartic proteases (Saps) that physically break down the blood-brain barrier, opening a pathway for the fungus to invade brain tissue.
“Our first question was, how does C. albicans enter the brain? We found that C. albicans produces enzymes called secreted aspartic proteases (Saps) that break down the blood-brain barrier, giving the fungus access to the brain where it causes damage,” said Dr. Yifan Wu, postdoctoral scientist at Baylor College of Medicine.
Another pathway involves Malassezia, a common skin fungus. A 2024 PubMed review found that Malassezia DNA has been detected in brain tissue and linked to Alzheimer’s, Parkinson’s, multiple sclerosis, and ALS.
Its ability to survive in the lipid-rich environment of brain cells — the same fatty environment that makes it thrive on human skin — may explain why it can persist in neural tissue.
Cryptococcus neoformans, a well-studied fungal pathogen, is known to enter the brain within 24 hours of infection and can remain dormant for extended periods while microglia (the brain’s immune cells) delay their response.
The Amyloid Connection: Your Brain’s Antifungal Defense
This is where the science becomes truly remarkable — and turns one of Alzheimer’s most debated theories upside down.
For years, the leading explanation for Alzheimer’s was the amyloid cascade hypothesis: toxic protein fragments called amyloid-beta (Aβ) build up in the brain, form plaques, and kill neurons.
The assumption was that this buildup was a malfunction — a disease process.
But researchers at Massachusetts General Hospital and Harvard Medical School proposed a radically different interpretation.
They found that amyloid-beta is actually an antimicrobial peptide — a natural defense weapon the brain deploys against invading fungi and bacteria.
In their 2018 paper in the journal Neuron, they showed that Aβ oligomers bind to microbial surface particles and trap pathogens in fibrillar nets. In other words, amyloid plaques may originally form as the brain’s attempt to cage an infection.
Research published in PMC in 2022 expanded on this model, explaining that Aβ acts through two direct pathways: binding to microbial surface carbohydrates to prevent fungi from spreading, and fibrillizing on microbial surfaces to rupture their membranes.
The problem arises when this defense process becomes chronic and turns on the brain itself — destroying the neurons it was meant to protect.
The Baylor College of Medicine team confirmed this in animal models: Candida albicans infection in the brain directly generates amyloid-beta-like peptides — toxic protein fragments that are the hallmark of Alzheimer’s disease pathology.
“We propose that the brain Aβ-peptide aggregates that characterize multiple Candida-associated neurodegenerative conditions including Alzheimer’s disease, Parkinson’s disease and others, may be generated both intrinsically by the brain and by C. albicans,” said Dr. David Corry, Fulbright Endowed Chair in Pathology at Baylor College of Medicine.
Mycotoxins: The Chemical Assault on the Brain
Even without a direct fungal infection in the brain, the toxic chemicals that molds produce — mycotoxins — can travel from the respiratory system into the bloodstream and cross the blood-brain barrier on their own.
An earlier but often-cited study published in Toxicology and Industrial Health established the clinical framework that continues to inform modern research.
The study documented that human exposure to molds, mycotoxins, and water-damaged buildings can cause neurologic and neuropsychiatric signs and symptoms, and that “mycotoxins can affect sensitive individuals, and possibly accelerate underlying neurologic/pathologic processes”.
A 2025 study published in Toxicology examined the mechanisms by which mycotoxin exposure links to neurodegenerative diseases like Alzheimer’s and Parkinson’s.
Researchers identified multiple attack pathways:
Oxidative stress: Mycotoxins generate reactive oxygen species (ROS) that damage neurons and deplete antioxidant defenses
Mitochondrial dysfunction: Mycotoxins impair the energy-producing organelles inside cells, starving neurons
Neuroinflammation: Mycotoxins activate NLRP3 inflammasomes in microglia, triggering chronic brain inflammation
BBB disruption: Compounds like aflatoxin B1 reduce key transport proteins, making the brain more permeable to toxins
Direct neurotoxicity: Several mycotoxins — including ochratoxin A, fumonisin B1, and trichothecenes — have been directly detected in brain tissue
Satratoxins produced by Stachybotrys chartarum (black mold) are especially destructive. Research found that satratoxin causes apoptosis (programmed cell death) in the olfactory nerve layer of the brain and leads to bilateral atrophy. The same toxins were found to trigger deposition of amyloid-beta plaques in the frontal cortex — the same hallmark seen in Alzheimer’s pathology.
A 2023 review in IMR Press confirmed: “Inflammation stimulated by mycotoxins causes inflammatory markers such as NF-kappa B and TNF-alpha to access the OB and frontal cortex, leading to the deposition of amyloid-beta plaques (an Alzheimer disease pathological hallmark) and other neurological disorders.”
Decades of Drug Failure
For most of the last 20 years, the pharmaceutical response to Alzheimer’s has been a graveyard of failed promises. Hundreds of clinical trials. Billions of research dollars. And almost nothing to show for it.
The drugs that have existed since the late 1990s — cholinesterase inhibitors like donepezil, and memantine — offer modest, temporary symptom stabilization at best. They do not slow the disease. They do not stop the plaques from building. They do not halt the dying of neurons. They buy time, sometimes, in small amounts.
Between 2003 and 2022, over 99% of Alzheimer’s drug trials failed to produce meaningful results.
Theory after theory collapsed in clinical testing.
The amyloid hypothesis — the idea that clearing sticky amyloid plaques in the brain would halt the disease — was debated for years as researchers watched drug after drug fail even when it successfully cleared the plaques.
The brain damage, it turned out, may begin long before any drug could intervene.
Conclusion
The idea that fungi/molds — some of the most common organisms on Earth — may be a driving force behind one of medicine’s most devastating diseases is no longer a fringe theory.
It is supported by peer-reviewed research, confirmed in human brain tissue, replicated in animal models, and actively funded by major scientific institutions.
In this article, I show that for well over 20 years, multiple studies have shown that fungi/molds play a critical role in dementia and alzheimer’s disease and are also found in 100% of the brain tissues of these patients.
Yet, as of 2026, no antifungal drugs and or treatments addressing this known cause has been developed.
The disease continues to wreak havoc on the world.
Over the last 20 years, the disease has quietly become one of the most devastating health crises in modern history — and the worst is still ahead.
Deaths tell an even darker story. Between 2000 and 2021, while deaths from heart disease, stroke, and HIV were declining, Alzheimer’s deaths climbed more than 140%.
An estimated 7.2 million Americans age 65 and older live with Alzheimer’s dementia today, with projections suggesting that number could reach 13.8 million by 2060.
The number of Americans developing dementia each year is projected to grow from 514,000 in 2020 to 1 million per year by 2060.
These diseases went in the opposite direction of nearly every other major killer in America.
They didn’t shrink.
They exploded…
And the lifetime risk figures are worse than most people realize.
A landmark 2025 NYU Langone study concluded that the lifetime risk of developing dementia after age 55 is now 42% — more than double what older studies suggested.
Nearly half of the US population are predicted to develop dementia after 55!
The cost of all this suffering?
In 2023 alone, unpaid family caregiving for dementia patients was valued at $346.6 billion.
That doesn’t count the lost careers, the broken marriages, the children watching their parents disappear piece by piece.
This number was hidden for years because earlier research quietly undercounted mild and early-stage cases.
What this tells us is that brain health cannot be separated from environmental health.
The air we breathe inside our homes, the moisture levels in our walls, and the invisible biological agents that colonize water-damaged buildings may have consequences that extend far beyond respiratory symptoms.
They may reach the neurons responsible for memory, identity, and cognition itself.
For homeowners, renters, and property managers, the message is clear: mold is not just a cosmetic problem or an air quality inconvenience.
It is not only a potential neurological hazard, it is possibly causes some of the most horrific diseases that humans are suffering from today like dementia and alzheimer’s.
Early detection, proper remediation, and awareness of fungal exposure history are tools that may — over time — prove as important to dementia prevention as any pharmaceutical intervention yet developed.
References
-
Pisa, D., et al. (2015). “Different Brain Regions are Infected with Fungi in Alzheimer’s Disease.” Scientific Reports, Nature. https://www.nature.com/articles/srep15015
-
Pisa, D., et al. (2018). “Infection of Fungi and Bacteria in Brain Tissue From Elderly Persons and Patients With Alzheimer’s Disease.” Frontiers in Aging Neuroscience. https://pmc.ncbi.nlm.nih.gov/articles/PMC5976758/
-
Wu, Y., et al. (2023). “Brain fungal infection produces Alzheimer’s disease-like changes.” Baylor College of Medicine. Cell Reports. https://www.bcm.edu/news/brain-fungal-infection-produces-alzheimers-disease-like-changes
-
Moir, R.D., et al. (2018). “Alzheimer’s Disease-Associated β-Amyloid Is Rapidly Seeded by Herpesviridae to Protect against Brain Infection.” Neuron. https://pubmed.ncbi.nlm.nih.gov/30001512/
-
From Skin and Gut to the Brain: The Infectious Journey of Malassezia. (2024). PubMed. https://pubmed.ncbi.nlm.nih.gov/38871941/
-
A potential etiological factor for neurodegenerative diseases — Mycotoxins. (2025). ScienceDirect/Toxicology. https://www.sciencedirect.com/science/article/abs/pii/S0300483X25000125
-
Mycotoxins Exposure: Neuroinflammation, Cognitive Decline and Neurodegenerative Diseases. (2025). Lidsen Neurobiology. https://www.lidsen.com/journals/neurobiology/neurobiology-09-04-305
-
Gut Fungi in Alzheimer’s Disease. (2025). Aging and Disease. https://www.aginganddisease.org/EN/10.14336/AD.2024.1310
-
Mold and Mycotoxin Exposure and Brain Disorders. (2023). IMR Press. https://www.imrpress.com/journal/JIN/22/6/10.31083/j.jin2206137
-
Innate Immune and Fungal Model of Alzheimer’s Disease. (2018). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC6159659/
-
Infectious Origin of Alzheimer’s Disease. (2022). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC9671327/
-
Neurologic and Neuropsychiatric Syndrome Features of Mold and Mycotoxin Exposure. (2009). PubMed/JOEM. https://pubmed.ncbi.nlm.nih.gov/19854819/
-
IDSA Microbial Pathogenesis in Alzheimer’s Disease Research Grant. https://www.idsociety.org/practice-resources/grants-and-funding/microbial-pathogenesis-in-alzheimers-disease-research-grant-program
-
Fungal Infections, Use of Antifungal Agents, and the Risk of Alzheimer’s Disease. (2022). Oxford Academic / Innovation in Aging. https://academic.oup.com/innovateage/article/6/Supplement_1/1/6937060
- How a Slime Mold Came to the Aid of Alzheimer’s Research by Kathleen Cason


0 Comments