Semen (seminal fluid) is not sterile.
It contains a rich and diverse community of microorganisms, including bacteria, viruses, and fungi, some of which can cause real harm to both male and female reproductive health.
Peer-reviewed studies from multiple countries have detected live fungal organisms, including Candida albicans, Candida glabrata, Candida parapsilosis, and even Cryptococcus neoformans, inside human seminal fluid.
A landmark study published in Fertility and Sterility found that most semen samples contain 28% of all detectable fungal growth, with Candida species being the most prevalent isolates.
Fungi in seminal fluid can include Candida albicans, Candida guilliermondii, Candida parapsilosis, and Candida glabrata, detected at concentrations ranging from 10² to over 10³ colony-forming units per milliliter.
A 1975 study published in PubMed found Candida albicans isolated from patients with prostatitis symptoms, appearing more frequently after antibiotic treatment (24%) than before (15%), suggesting that disrupting the bacterial microbiome may allow fungal overgrowth.
A critical but underappreciated case involves Cryptococcus neoformans, a serious fungal pathogen: research has documented its persistence in both seminal fluid and urine even during itraconazole treatment, identifying the prostate gland specifically as a long-term reservoir where it can survive antifungal therapy.
Semen itself also appears to actively support fungal growth.
A 2020 study found that semen medium sustained Candida albicans growth, functioning as a culture medium that allowed the yeast to proliferate, though not as robustly as standard laboratory media.
Research published in PubMed examining urogenital tract infections found that Candida albicans appeared in prostatitis patients at a rate of 15% before antibiotic treatment and 24% after antibiotic treatment — a statistically significant increase that suggests disrupting the bacterial microbiome through antibiotic use can allow fungal organisms to fill the ecological gap left behind.
Perhaps the most striking fungal reservoir in male reproductive anatomy is the prostate gland. A study on Cryptococcus neoformans in reproductive fluids concluded:
“The prostate gland may serve as a sanctuary site where Cryptococcus neoformans can persist despite systemic antifungal therapy, allowing viable organisms to be shed continuously into seminal fluid and urine.”
This finding carries serious clinical implications.
A man undergoing treatment for cryptococcal infection may still harbor viable fungal cells in his prostate — and shed them in every ejaculation — even while appearing to respond to treatment.
Aspergillus species have also been documented infecting the male urogenital system, including the penis, scrotum, and testes, though these cases are typically seen in immunocompromised patients.
How Fungi Travel in Semen: The Biology of Transmission
Fungi access seminal fluid through multiple pathways. The urethra carries commensal organisms that mix with ejaculate during passage.
The prostate, which contributes roughly 30% of seminal fluid volume, can shed fungal colonies directly into the ejaculate. The seminal vesicles and epididymis also contribute fluids that may carry organisms from surrounding tissue.
Once inside semen, fungi do not simply float passively.
Research has shown that Candida albicans actively attaches to spermatozoa, using the sperm head as a primary binding site.
A 2007 study published in the International Journal of Andrology investigated the direct interaction between Candida albicans and human sperm cells, concluding:
“Candida albicans cells adhered primarily to the sperm head region, and this attachment was associated with significant impairment of sperm motility and morphological changes to the sperm ultrastructure.”
The damage is not limited to physical attachment.
Candida species also release soluble metabolic factors — compounds secreted into the surrounding fluid — that inhibit sperm function even when no direct cell contact occurs.
A 2018 study in PMC on the effects of Candida on semen found that both C. albicans and C. glabrata filtrates significantly reduced sperm motility and forward progression. The researchers stated:
“The deleterious effect of Candida spp. on spermatozoa involves not only direct contact mechanisms but also the secretion of soluble factors including farnesol and other quorum-sensing molecules that cause measurable deterioration of sperm functional parameters.”
Semen itself also appears to actively nurture fungal growth. A 2020 study published in the Journal of Obstetrics and Gynaecology Research found that semen medium supported the proliferation of Candida albicans, essentially functioning as a growth medium. The researchers noted:
“Semen supported the growth of Candida albicans, though not as effectively as synthetic laboratory media. This property of semen may represent a biological risk factor for male-mediated transmission of vaginal candidiasis.”
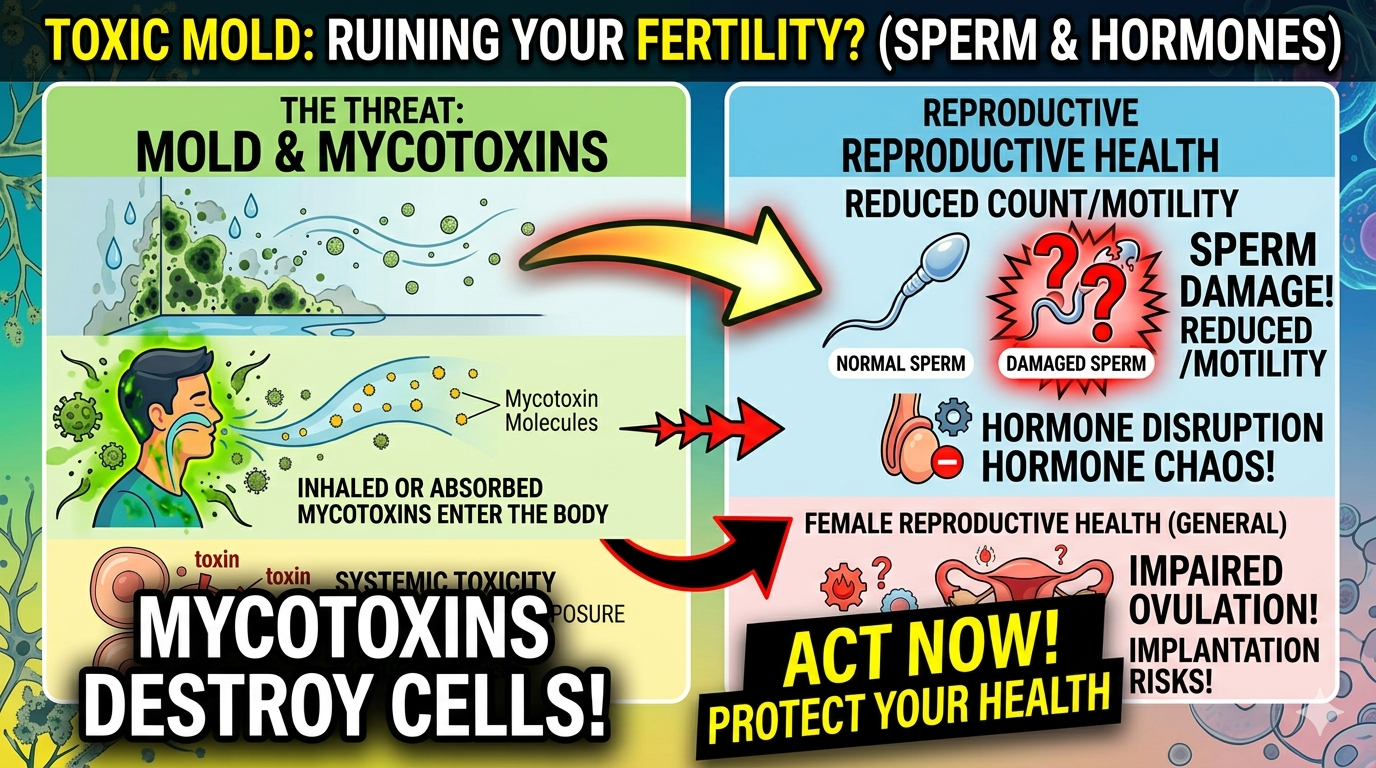
Mold Mycotoxins and Sperm Quality
There is a related but distinct dimension to this topic: the impact of environmental mold mycotoxins on seminal quality.
Men who are exposed to mold toxins through contaminated indoor air or food can absorb those toxins systemically, and those toxins can appear in reproductive tissues and fluids.
A 2024 study published in PubMed and PMC assessed urinary levels of mycotoxins (zearalenone, ochratoxin A, and deoxynivalenol) in 136 infertile men versus 154 fertile controls and found that mycotoxin exposure reduces sperm motility and morphology.
The results were striking. The researchers concluded:
“Mycotoxin exposure was significantly associated with reduced sperm motility, abnormal morphology, and decreased chromatin integrity. These effects are likely mediated through mycotoxin-induced endocrine disruption, oxidative stress, and mitochondrial dysfunction in spermatozoa.”
A 2024 study in Science of the Total Environment further evaluated the joint exposure to a spectrum of mycotoxins and its association with semen quality, adding to the growing evidence that mold exposure has direct consequences for male reproductive health.
The Infertility Connection
The reproductive consequences of fungal semen contamination go beyond infection transmission. Research shows that fungal organisms in semen attack sperm function through multiple pathways:
Motility: Candida species directly inhibit the swimming ability of sperm, reducing forward progression
Morphology: Structural abnormalities in sperm heads and tails are associated with C. albicans and C. glabrata exposure
Viability: Studies confirm that experimental Candida infection reduces sperm’s fertilizing ability in assisted reproduction models
DNA integrity: Mycotoxins reduce chromatin stability, potentially contributing to embryo quality issues
Genitourinary tract infections account for approximately 15% of male infertility cases, with fungal infections being significantly underdiagnosed compared to bacterial causes.
As researchers at Frontiers in Immunology noted in 2023, the effect of fungal and viral infections on male fertility is commonly undervalued relative to bacterial infections, despite evidence of clear harm.
Conclusion
Semen is a biologically complex fluid that can transport live fungi from the male urogenital tract with real consequences for both partners’ health and for reproductive outcomes.
Candida species are present in roughly one in four semen samples in some clinical populations, where they damage sperm and facilitate female partner reinfection.
Cryptococcus neoformans can persist as a long-term prostatic reservoir, escaping treatment while continuing to contaminate ejaculate.
Separate from direct fungal transmission, systemic exposure to mold mycotoxins from contaminated indoor environments can silently degrade sperm quality through endocrine disruption, oxidative stress, and DNA damage.
These two pathways — internal fungal colonization and external environmental mycotoxin exposure — can converge, creating compounded reproductive harm that is rarely addressed in conventional infertility evaluations.
The message for clinicians, patients, and mold investigators is the same: fungi are not peripheral to reproductive health. They sit at its center, and the evidence is there for those willing to look.
References
-
Mårdh, P.A. et al. (1975). Search for uro-genital tract infections in patients with symptoms of prostatitis. PubMed. https://pubmed.ncbi.nlm.nih.gov/175434/
-
Maduna, L.D. et al. (2020). Mycological Analysis for Seminal Fluid before In Vitro Fertilization Procedure. EIJPPR. https://eijppr.com/article/mycological-analysis-for-seminal-fluid-before-in-vitro-fertilization-procedure
-
Espinosa-Hernández et al. (2018). Yeast and Fertility: Effects of In Vitro Activity of Candida spp. on Semen. PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC5960052/
-
Rennemeier, C. et al. (2007). Candida albicans and filtrates interfere with human spermatozoal motility. Wiley. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2605.2006.00734.x
-
Spinillo, A. et al. (1992). Recurrent vaginal candidiasis. PubMed. https://pubmed.ncbi.nlm.nih.gov/1593559/
-
Persistence of Cryptococcus neoformans in seminal fluid. PubMed. https://pubmed.ncbi.nlm.nih.gov/1965324/
-
Microbiota of the seminal fluid from healthy and infertile men. PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC3888793/
-
Association of Urinary Mycotoxins with Sperm Quality. PMC/NIH. https://pmc.ncbi.nlm.nih.gov/articles/PMC10974026/
-
Semen supports growth of Candida albicans. BJOG. https://obgyn.onlinelibrary.wiley.com/doi/abs/10.1111/jog.14367
-
Sperm immobilization factor of Candida albicans. Reproductive Group. https://www.reprodgroup.us/articles/GJFR-5-114.php
-
Recent Semen Exposure Impacts Cytokine Response. PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC8221111/
-
Aspergillosis of the Genito-Urinary Tract. Clinic Search Online. https://www.clinicsearchonline.org/article/aspergillosis-of-the-genito-urinary-tract-and-kidney-including-the-penis-scrotum-test


0 Comments